Our Products
GH2S is – Green Hydrogen system use hydrogen gas as fuel to generate electrical energy.
What is GH2S?
GH2S is – Green Hydrogen system – System use hydrogen gas as fuel to generate electrical energy where green is the method of producing hydrogen. By using Reliable energy to perform the is method of producing hydrogen Call Green Hydrogen. Hydrogen is one of the most available elements in the universe but hydrogen dose not found alone as gas, Usually found combine with other elements like CH4 (Methan), H2O (Water),…..etc. The most common shape of finding Hydrogen is water as water is every wear, spiting Hydrogen from Oxygen is process call electrolyzing. Electrolyzer is Device cathode (Negative Charge) and anode (Positive Charge) to Split the water into Hydrogen gas and Oxygen gas. After Hydrogen is spit from oxygen it stores in storage tanks for further usage. Finaly Fuel Cell are used to convert hydrogen gas to Electrical Energy.

What is the difference between Green Blue and grey Hydrogen?
The terms “Green,” “Gray,” and “Blue” hydrogen refer to different methods of producing hydrogen, each with distinct environmental and economic characteristics. These terms are often used to describe the carbon footprint associated with the hydrogen production process.
- Green Hydrogen:
- Production Method: Green hydrogen is produced using renewable energy sources, typically through the process of electrolysis, where water is split into hydrogen and oxygen using electricity generated from renewable sources like solar or wind power.
- Environmental Impact: Green hydrogen is considered environmentally friendly because it has a minimal carbon footprint. The production process releases only water vapor as a byproduct, making it a clean and sustainable option.
- Gray Hydrogen:
- Production Method: Gray hydrogen is produced through a process called steam methane reforming (SMR), which involves extracting hydrogen from natural gas. This is the most common method of industrial hydrogen production but is not environmentally friendly.
- Environmental Impact: The production of gray hydrogen involves releasing carbon dioxide (CO2) as a byproduct, contributing to greenhouse gas emissions. It is considered less environmentally sustainable compared to green hydrogen.
- Blue Hydrogen:
- Production Method: Blue hydrogen is produced using the same methods as gray hydrogen, primarily steam methane reforming. However, the key difference is that the carbon emissions generated during production are captured and stored using carbon capture and storage (CCS) technologies to prevent them from entering the atmosphere.
- Environmental Impact: Blue hydrogen aims to mitigate the environmental impact associated with gray hydrogen by capturing and storing the carbon emissions. While it is not as clean as green hydrogen, it is considered a transitional option that reduces the overall carbon footprint of hydrogen production.
In summary, the color classification of hydrogen—green, gray, or blue—reflects the environmental considerations associated with its production method. Green hydrogen is produced using renewable energy and has a minimal carbon footprint, while gray hydrogen is produced from natural gas without carbon capture, contributing to greenhouse gas emissions. Blue hydrogen is produced similarly to gray hydrogen but incorporates carbon capture and storage to reduce its environmental impact
What is the Parts of GH2S?
Energy, Water, Electrolyzer, Fuel Cell, Storage Tanks.
Reliable energy Source:

Wind, Solor, nuclear, Solor Energy is the most recommended Source to be use to be used for this system as whereever there is sun Solor energy can be found.
Water:

Electrolyzer
Electrolyzer is the Device that use Electrical Power to split water into Hydrogen gas And Oxygen gas. Electrcolyzer have two main part cathode (Negative Charge) and anode (Positive Charge) which will be submerge in water, Cathode (-) pulls Hydrogen (+) And Anode (+) pulls Oxygen atoms (-). At the End Two Gases will be the produces.

Fuel Cell:
A fuel cell is an electrochemical device that converts chemical energy directly into electrical energy. It does so through an electrochemical reaction between a fuel, typically hydrogen, and an oxidizing agent, usually oxygen from the air. The most common type of fuel cell is the proton exchange membrane (PEM) fuel cell.
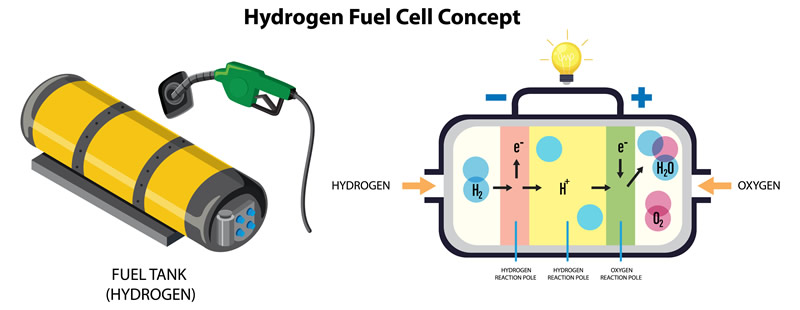
Fuel cells are considered a clean and efficient energy technology because they produce electricity without combustion, and the only byproducts are water vapor and heat. They are often used in various applications, including stationary power generation, transportation (such as fuel cell vehicles), and portable electronic devices. Fuel cells offer the potential for lower emissions and greater energy efficiency compared to traditional combustion-based power systems.

